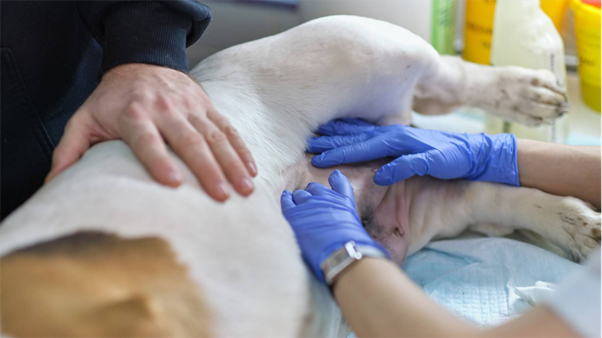
Article
Uterine Biopsy in the Subfertile Bitch: Moving from Theory to Clinical Decision-Making
Endometrial biopsy has long been a cornerstone in evaluating reproductive health in large animals, particularly in species like cows and mares, where it directly correlates with fertility outcomes. However, its application in canine practice remains less clearly defined. For veterinary clinicians dealing with subfertility in bitches, uterine biopsy presents both an opportunity and a challenge, offering histological insights while still lacking fully established prognostic benchmarks1.
Lessons from Large Animal Practice
In large animal reproduction, uterine biopsy is not merely diagnostic but prognostic. In mares, biopsy grading directly reflects the ability to carry a pregnancy to term. Parameters such as inflammation, periglandular fibrosis, and fibrotic nests are routinely assessed, making biopsy an integral part of reproductive soundness evaluation. Similarly, in dairy cows, endometrial biopsy enables early and sensitive detection of endometritis, helping identify animals requiring treatment before breeding1,2.
This structured approach highlights what canine practice currently lacks, a standardized interpretation system that directly links histopathology with reproductive outcomes.
The Clinical Need in Canine Practice
Unlike polyestrous species, the domestic dog is monoestrous with limited reproductive opportunities. This makes each breeding attempt valuable, both economically and genetically. Repeated failed breeding attempts in subfertile bitches result in significant losses, emphasizing the need for more definitive diagnostic tools1.
Uterine biopsy has been explored as one such tool, particularly to evaluate lesions like cystic endometrial hyperplasia (CEH) and endometritis. However, a major limitation in the literature has been the lack of direct comparison between fertile and subfertile bitches, an essential step for meaningful clinical interpretation.
Biopsy Techniques and Practical Considerations
Two primary techniques are described in canine patients1:
- Full-thickness biopsy via hysterotomy
- Partial-thickness biopsy using transcervical forceps
While both methods are technically feasible, their diagnostic yield may differ. Full-thickness biopsies, although invasive, allow evaluation beyond the endometrium into deeper structures such as the myometrium. This becomes clinically significant when lesions are not confined to superficial layers.
What the Evidence Suggests
Recent comparative findings provide valuable clinical direction. Among the evaluated parameters, inflammation, fibrosis, and endometrial cysts, only inflammation showed a statistically significant difference between fertile and subfertile bitches (p=0.004). In contrast, fibrosis and cysts did not demonstrate significant differences (p>0.05)1.
This is particularly relevant for clinicians who may overinterpret mild fibrosis or incidental cysts. While fibrosis is a known fertility-limiting factor in mares, similar conclusions cannot be directly applied to dogs. Mild fibrosis may not impair fertility in the bitch, likely due to differences in placentation mechanisms1,3.
Rethinking the Role of Cysts
Cystic endometrial hyperplasia is frequently reported in canine uterine pathology and is associated with conditions like pyometra1. However, its presence alone may not indicate subfertility. Comparable incidence rates in fertile and subfertile bitches suggest that cysts, especially when isolated, should not be overemphasized during clinical decision-making.
This reinforces the importance of correlating histopathology with clinical history and imaging findings, rather than relying on biopsy in isolation1.
Inflammation: The Key Diagnostic Indicator
Among all parameters, inflammation, specifically lymphoplasmacytic inflammation, emerges as the most clinically relevant finding. Its significantly higher prevalence in subfertile bitches (p<0.001) suggests a strong association with impaired fertility1.
Importantly, this form of inflammation may indicate a chronic disease process, particularly when observed in younger animals. This shifts the clinical focus toward identifying and managing chronic uterine pathology rather than transient inflammatory changes.
Practical Takeaways
For practicing veterinarians, several actionable insights emerge:
- Do not overinterpret mild fibrosis or isolated cysts in biopsy reports
- Prioritize identification and characterization of inflammation, especially lymphoplasmacytic types
- Consider full-thickness biopsy when deeper pathology is suspected
- Always interpret biopsy findings in conjunction with reproductive history and clinical context
Conclusion
Uterine biopsy in the bitch is evolving from a purely descriptive tool to a more clinically meaningful diagnostic aid. While limitations remain, particularly in standardization, current evidence highlights inflammation, rather than fibrosis or cysts, as the most relevant indicator of subfertility.
As further research refines biopsy interpretation, veterinarians can increasingly rely on this tool to guide breeding decisions, optimize reproductive outcomes, and minimize economic losses in canine practice.
References:
- McRae GR, Coutinho da Silva MA, Runcan EE, Stephens JA, Premanandan C. Histological Studies in the Endometrium of Fertile and Subfertile Bitches. Reproduction in Domestic Animals. 2025 Apr;60(4):e70055. https://onlinelibrary.wiley.com/doi/pdf/10.1111/rda.70055
- Westendorf, J., B. Wobeser, and T. Epp. 2022. “IIB or Not IIB, Part 1: Retrospective Evaluation of Kenney-Doig Categorization of Equine Endometrial Biopsies at a Veterinary Diagnostic Laboratory and Comparison With Published Reports.” Journal of Veterinary Diagnostic Investigation 34, no. 2: 206–214. https://journals.sagepub.com/doi/pdf/10.1177/10406387211062207
- Premanandan, C., and E. Runcan. 2019. “Canine Placentation: Normal Gross and Histologic Structure, and Confounding Features of Evaluation.” Clinical Theriogenology 11, no. 4: 623–625. https://clinicaltheriogenology.net/index.php/CT/article/download/9463/15358
Related Contents
Article
The Diagnostic Discipline of Pruritus – Why Structure Beats Guesswork
Pruritus remains one of the most common presenting complaints in small animal practice, ye...
Article
Feline Allergic Dermatitis — A Practical, Stepwise Diagnostic Approach
Once the complexity of feline allergic dermatitis is understood, the solution lies in discipline. Th...
Article
Feline Allergic Dermatitis — Why Diagnosis is So Challenging
Feline allergic dermatitis does not follow predictable rules—and that is exactly why it is so...

Article
Food Allergies in Cats – Managing the Fussy Eaters
Food allergies are a common, yet often under-recognized cause of chronic pruritus in cats. Unlike do...
.jpg)
Article
Feline Atopic Skin Syndrome (FASS) – Beyond the Itch
Feline Atopic Skin Syndrome (FASS) is a chronic, inflammatory skin condition triggered by...
Article
Allergic Otitis Externa in Dogs and Cats: Diagnosis and Management
Allergic otitis externa (AOE) is a frequent manifestation of allergy in companion animals...

Article
Spectrum of Care Considerations in Managing Allergic Skin Diseases
Allergic skin diseases are among the most common medical conditions in dogs and cats, with skin alle...

Article
Understanding Uterine Lesions in Canine Subfertility: What Really Matters Clinically?
Subfertility in the bitch is a multifactorial problem, often frustrating for both clinicians and bre...